Welcome to the Huang Research Group!
Huang’s group explores the unique technological opportunities that result from the structure and assembly of nanoscale building blocks. Taking advantage of the unique roles of nanoscale surfaces and interfaces, our group is creating methodologies to apply the latest developments in nanoscale materials and nanotechnology for probing nanoscale processes that can fundamentally impact a wide range of technologies including materials synthesis, catalysis, fuel cells, and devices applications.
Group News
-
Read more...
 Professor Yu Huang has been awarded the 2023 Eni Energy Transition Award for Innovation in Hydrogen Fuel Cells.
Professor Yu Huang has been awarded the 2023 Eni Energy Transition Award for Innovation in Hydrogen Fuel Cells.
 Prof. Huang is named among 2023 highly cited researchers by Clarivate in the past six consecutive years
Prof. Huang is named among 2023 highly cited researchers by Clarivate in the past six consecutive years
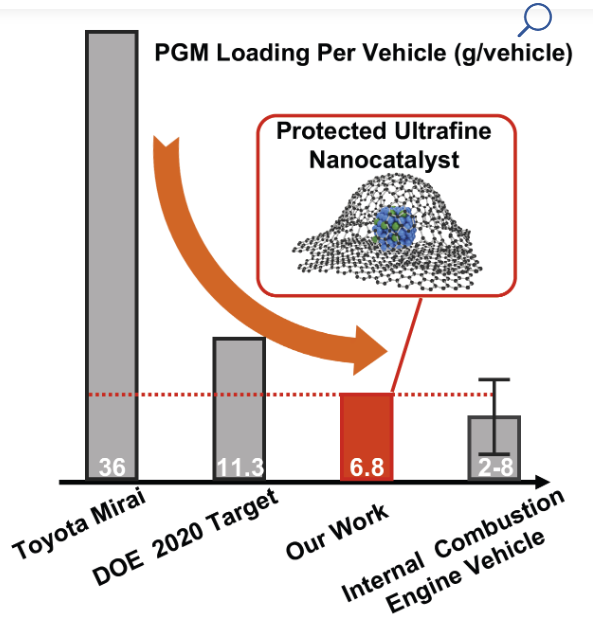 Hydrogen fuel cell advance: UCLA team exceeds DOE-set targets for fuel cell performance and durability
Hydrogen fuel cell advance: UCLA team exceeds DOE-set targets for fuel cell performance and durability
 Prof. Huang is named among 2022 highly cited researchers by Clarivate in the past five consecutive years
Prof. Huang is named among 2022 highly cited researchers by Clarivate in the past five consecutive years
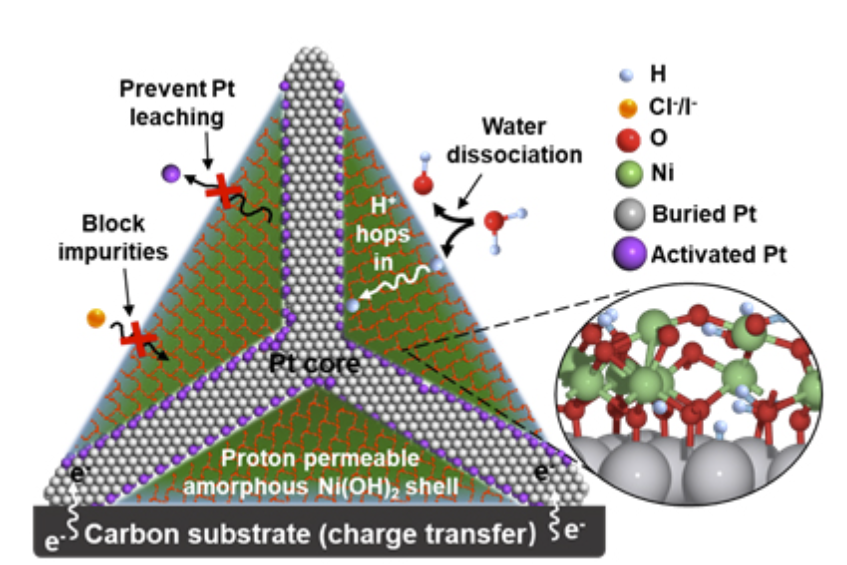 Bare-foot Ni(OH)2-clothed platinum-tetrapods enable more efficient green hydrogen production
Bare-foot Ni(OH)2-clothed platinum-tetrapods enable more efficient green hydrogen production
 Jin Huang achieved Department outstanding PhD Student and ACS ENFL future investigator award. Congratulations!
Jin Huang achieved Department outstanding PhD Student and ACS ENFL future investigator award. Congratulations!






